
Researchers at the Spanish National Cancer Research Centre (CNIO), led by Mariano Barbacid, developed a triple-drug therapy that completely eliminated pancreatic ductal adenocarcinoma (PDAC) tumors in three mouse models, with no recurrence observed for over 200 days and minimal side effects.
Study Overview
The research, published in Proceedings of the National Academy of Sciences (PNAS), targeted PDAC, the most common and lethal form of pancreatic cancer, which often develops resistance to single-drug treatments within months.After six years of work, the team achieved permanent tumor regression by simultaneously inhibiting three key signaling pathways downstream of the KRAS oncogene: RAF1, EGFR family receptors, and STAT3.
Therapy Components
- RMC-6236 (daraxonrasib): An experimental KRAS inhibitor already approved for lung cancer.
- Afatinib: An inhibitor of EGFR family receptors.
- SD36: A selective STAT3 protein degrader.
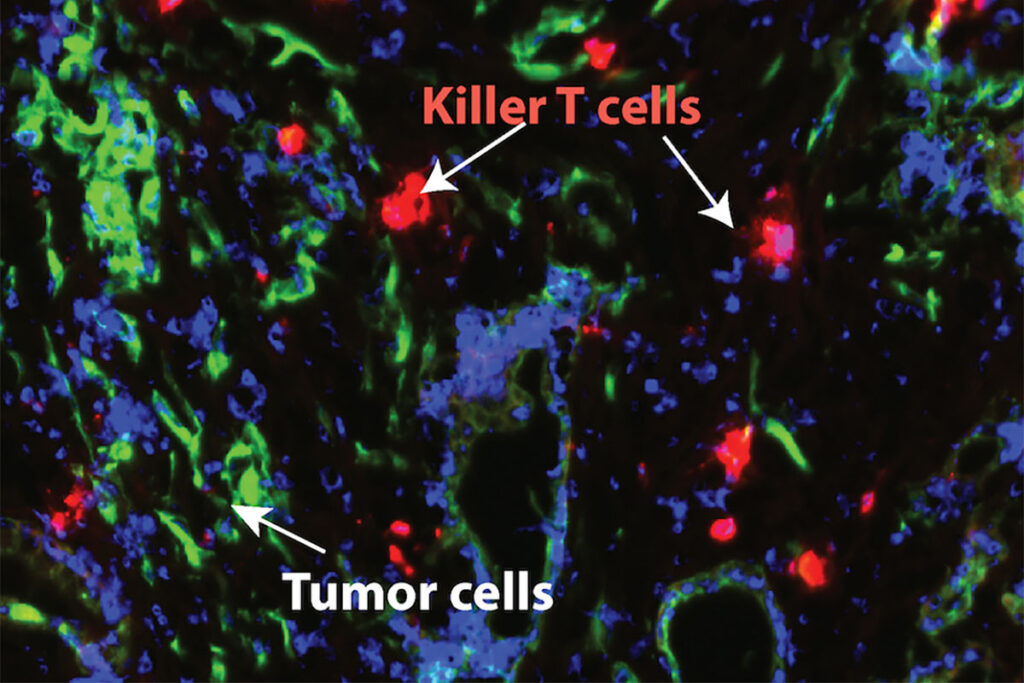
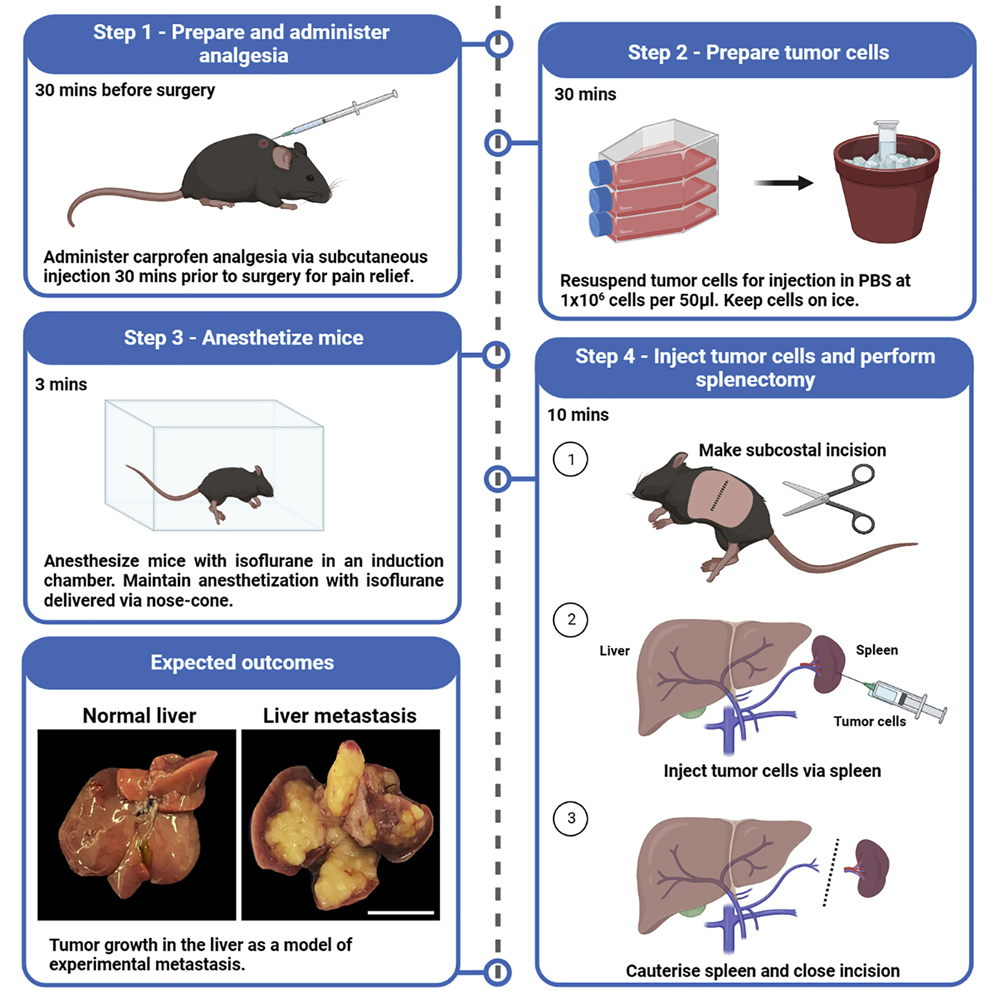
In orthotopic mouse models—where tumor cells are implanted in the pancreas to mimic the natural environment—the combination reduced tumors to undetectable levels without significant toxicity.
Key Findings
- Complete and permanent tumor disappearance in all tested models, overcoming resistance mechanisms that allow tumors to adapt and regrow.
- No evidence of relapse during extended monitoring (over 200 days).
- Minimal side effects, unlike many cancer therapies, making it promising for potential human use.
The authors noted: “These studies open a path to designing new combination therapies that can improve survival for patients with pancreatic ductal adenocarcinoma. These results point the way for developing new clinical trials.”
Implications and Limitations
This multi-pathway approach addresses PDAC’s adaptability, where single-target drugs fail due to tumor rewiring. Independent experts highlighted the rarity of durable responses without relapse in pancreatic cancer models.
However, the study is preclinical, limited to mice, and human clinical trials are not yet underway. The authors emphasized caution, as translation to humans requires further testing. The Spanish Embassy in the UK publicized the results on January 28, 2026, calling it a potential game-changer.

